Weizmann Institute of Sciences
Julian Savulescu, University of Oxford; Christopher Gyngell, The University of Melbourne, and Tsutomu Sawai, Hiroshima University
In the latest study, the scientists started with collections of stem cells. The conditions created by the external uterus triggered the developmental process that makes a fetus. Although the scientists said we are a long way off synthetic human embryos, the experiment brings us closer to a future where some humans gestate their babies artificially.
In the last few years, scientists have learned more about how to coax stem cells to develop into increasingly sophisticated structures, including ones that mimic the structure and function of human organs (organoids). Artificial human kidneys, brains, hearts and more have all been created in a lab, though they are still too rudimentary for medical use.
Another important legal issue is the source of stem cells and consent. The synthetic mouse embryo creators used stem cells from early embryos.
One of the key differences between organoids and synthetic embryos is their potential. If a synthetic embryo can develop into a living creature, it should have more protection than those which don’t.
These new technologies raise several ethical and legal concerns.
If you’re picturing a kind of silicone womb, think again. The external uterus is a rotating device filled with glass bottles of nutrients. This movement simulates how blood and nutrients flow to the placenta. The device also replicates the atmospheric pressure of a mouse uterus.
Artificial wombs
We should not let our fears about cloning stand in the way of crucial research. The benefits could make organ donor waiting lists a thing of the past, save premature babies and give women an option to have children a different way. Cloning, or any other unethical use of the technology, can be prevented by regulation.![]()
This article is republished from The Conversation under a Creative Commons license. Read the original article.
Each year over 300,000 women worldwide die in childbirth or as a result of pregnancy complications, many because they lack basic care. Even in wealthy countries, pregnancy and childbirth is risky and healthcare providers are criticised for failing mothers.
However, in the future it might be possible to make synthetic embryos from induced pluripotent stem cells (IPS). The worst case scenario would be a person donates a skin cell to research into producing organs to cure disease but this is used without their knowledge or consent to produce synthetic embryos.
Synthetic embryos and organs
In 2021 the research team used the same artificial womb to grow natural mouse embryos (fertilised from sperm and eggs), which lived for 11 days. The lab-created womb, or external uterus, was a breakthrough in itself as embryos could not survive in petri dishes.
IPS cells are created by taking a mature cell (such as a skin cell) from a living or dead person and applying treatments which drive it backwards to a more immature state. If the cell could be driven all the way back to an embryonic stem cell, it may one day be possible to use IPS cells to make viable embryos.
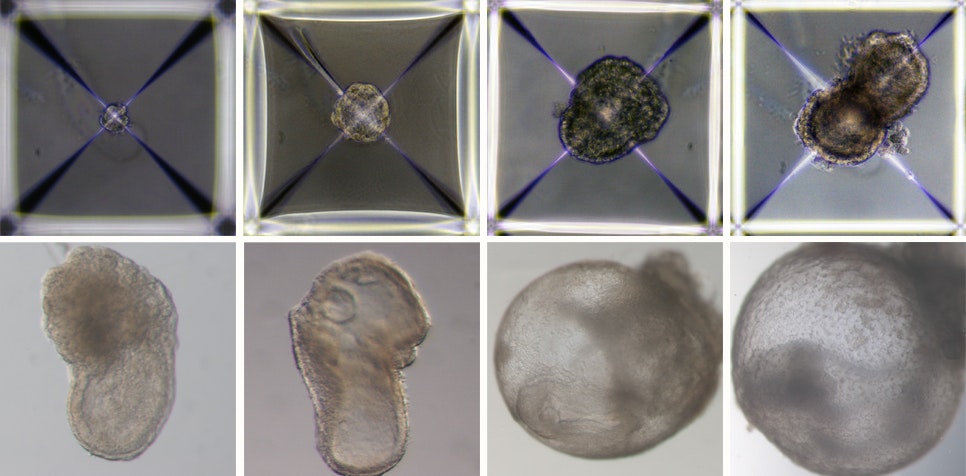
Some of the cells were treated with chemicals, which switched on genetic programmes to develop into placenta or yolk sac. Others developed into organs and other tissues without intervention. While most of the stem cells failed, about 0.5% were very similar to a natural eight-day-old embryo with a beating heart, basic nervous system and a yolk-sac.
Some philosophers say there is a moral imperative to develop artificial wombs to help remedy the unfairness of parenting roles. But other researchers say artificial wombs would threaten a women’s legal right to terminate a pregnancy.
There is an urgent need to make healthcare more accessible across the planet, provide better mental health support for mothers and make pregnancy and childbirth safer. In an ideal world every parent should expect excellent care in all aspects of motherhood. This technology could help treat premature babies and give at least some women a different option: a choice of whether to carry their child or use an external uterus.
That embryo would be a clone of the cell donor. The public and scientists have huge concerns about human cloning.
Synthetic embryos do not currently have potential to actually create a living mouse. If scientists did make human synthetic embryos, but without the potential to form a living being, they should arguably be treated similarly to organoids.
Cloning
It lived for eight days, about half a mouse’s gestation period, inside a bioreactor in the lab.
Julian Savulescu, Visiting Professor in Biomedical Ethics, Murdoch Children’s Research Institute; Distinguished Visiting Professor in Law, University of Melbourne; Uehiro Chair in Practical Ethics, University of Oxford; Christopher Gyngell, Research Fellow in Biomedical Ethics, The University of Melbourne, and Tsutomu Sawai, Associate Professor, Graduate School of Humanities and Social Sciences, Hiroshima University
Children, even some who are too young for school, know you can’t make a baby without sperm and an egg. But a team of researchers in Israel have called into question the basics of what we teach children about the birds and the bees, and created a mouse embryo using just stem cells.
But it has been possible to clone a human being using a different process called nuclear transfer, for 25 years. Nuclear transfer created Dolly the Sheep in 1997 and a monkey in 2018. In the late 90s and early 2000s, a flurry of laws introduced around the world successfully banned human cloning.
The issue of whether there are moral differences between using stem cells to produce models of human organs for research and using stem cells to create a synthetic embryo are already playing out in law courts.
Some countries (for example Australia) have taken the position that synthetic embryos such as “blastoids” (which resemble five-to-six-day-old embryos) should be treated like natural embryos, because of similarities in structure. Other countries (such as the UK, the US, Japan) treat synthetic embryos as different from embryos because they can’t currently produce a live baby.

